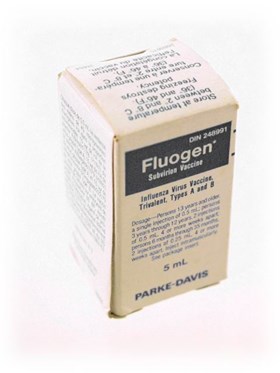
Influenza Virus Vaccine Trivalent, Types A and B [Fluogen®]
https://mhc.andornot.com/en/permalink/artifact4080
- Dates
- 1983
- circa 1983
- Collection
- Parke-Davis Collection
- Category
- Pharmacy and Drug Artifacts
- Vaccination
- Classification
- Pharmacy, Infectious Diseases
- Vaccination
- Accession Number
- 000001287 a-c
- Description
- Short cylindrical glass bottle (b) stopped with a rubber stopper and sealed with a metal cap moulded over the vial neck and lip with a plastic cap that is attached to the metal cap such that when the plastic cap is removed, the seal on the metal cap is broken; vial is in a cardboard box (a) with a …
1 image
- Accession Number
- 000001287 a-c
- Collection
- Parke-Davis Collection
- Category
- Pharmacy and Drug Artifacts
- Vaccination
- Classification
- Pharmacy, Infectious Diseases
- Vaccination
- MeSH Heading
- Viral Vaccines
- Vaccination
- Influenza
- MM= Drugs -- vaccines -- container
- MM= Drug Packaging -- container -- vial
- Description
- Short cylindrical glass bottle (b) stopped with a rubber stopper and sealed with a metal cap moulded over the vial neck and lip with a plastic cap that is attached to the metal cap such that when the plastic cap is removed, the seal on the metal cap is broken; vial is in a cardboard box (a) with a paper leaflet (c)
- Number Of Parts
- 3
- Part Names
- a - box - Size: Length 5.7 cm x Width 2.9 cm x Depth 2.9 cm
- b - vial - Size: Length 4.4 cm x Diam. 2.2 cm
- c - leaflet (unfolded) - Size: Length 17.6 cm x Width 10.1 cm
- Provenance
- Collected by Parke-Davis.
- Maker
- Parke-Davis Canada Inc.
- Dates
- 1983
- circa 1983
- Date Remarks
- Based on expiry date
- Material
- metal: silver
- paper: white; blue; black
- cardboard: white; blue; black
- rubber: pink
- glass: clear
- plastic: blue
- Inscriptions
- (a) Front: "DIN 248991 // Fluogen* // Subvirion Vaccine // Influenza Virus Vaccine. // Trivalent, Types A and B // Dosage – Persons 13 years and older, // a single injection of 0.5 mL; persons // 3 years through 12 years, 2 injections // of 0.5 mL, 4 or more weeks apart; // persons 6 months through 35 months, // 2 injections of 0.25 mL, 4 or more // weeks apart. Inject intramuscularly. // See package insert. // 5 mL // PARKE-DAVIS"; right side: "Contains no less than 45 micrograms // of HA antigen content per 0.5 mL // dose in the recemmended ratio of 15 // µg of HA of the A influenza virus // component represen- // tative of A/Bangkok/1/79 (H3N2) // and 15 µg of HA of the B influenza // virus component representative of // B/Singapore/222/79. The vaccine is / of chick embryo origin. Polysorbate // 80 NF, added during manufacture. // Preservative: 0.01% thimerosal (mer- // cury derivative). Standardized accord- // ing to the 1982-83 NACI and US // PHS requirements. // *Reg. T.M. / M. Enr. Parke, Davis & Company // Parke-Davis Canada Inc. // auth. user / usager aut. // Can. Lic. No. 1 // (L) 26227 // Exp. JN 1983 "; back: front repeated in French; left side: contents as printed on right side repeated in French; "Permis can. n° 1 // Parke-Davis Canada Inc. // Scarborough, Ontario // 266B670 26664"; top: "Store at temperature // between 2° and 8° C // (36° and 46° F) // Freezing destroys // potency. // [above 5 lines repeated in French] // 266B670 26664"; (b) front: "DIN 248991 // Fluogen* // Subvirion Vaccine // Influenza Virus Vaccine, // Trivalent, Types A and B // 5 mL // PARKE-DAVIS"; back: "(L) 26227 // EXP JN 1983 [stamped] // [contents, dosage and storage in English as on box] // Can. Lic. No. 1 // *Reg. T.M. Parke, Davis & Company // PARKE-DAVIS Canada Inc. auth. user // 266H130 26664"; bottom: "W // 37 [embossed]"; top: "FLIP // OFF [embossed]" (c) FLUOGEN* // INFLUENZA VIRUS VACCINE, TRIVALENT, TYPES A AND B // Subvirion Vaccine/Immunizing Antigen. // Ether Extracted // DESCRIPTION // [text follows] // CLINICAL PHARMACOLOGY // [text follows] // // 1 // [text continues from Clinical . . .] // INDICATIONS AND USAGE // [text follows] // CONTRAINDICATIONS // [text follows] // 2 // [text continues from Contraindications] // WARNING // [text follows] // PRECAUTIONS // [text follows] // PREGNANCY // [text follows] // ADVERSE REACTIONS // [text follows] // 3 // [text continues from Adverse . . .] // DOSAGE AND ADMINISTRATION // [text follows] // PACKAGE INFORMATION // [text follows] // REFERENCES // [text follows] // *Reg. T.M. / M. Enr. Parke, Davis & Company // Parke-Davis Canada Inc. auth. user / usager aut. // PARKE-DAVIS // 266E940 Parke-Davis Canada Inc., Scarborough, Ontario 26664 // 4 // [French translation occurs alongside English text throughout]"
- Permanent Location
- Storage Room 0010
- 0010-A3-5 Row G
- Condition Remarks
- Box somewhat dirty, dented; no apparent problems with vial or leaflet
- Copy Type
- Original
- Research Facts
- This is a vaccine against the influenza virus and is administered intramuscular with a hypodermic syringe.